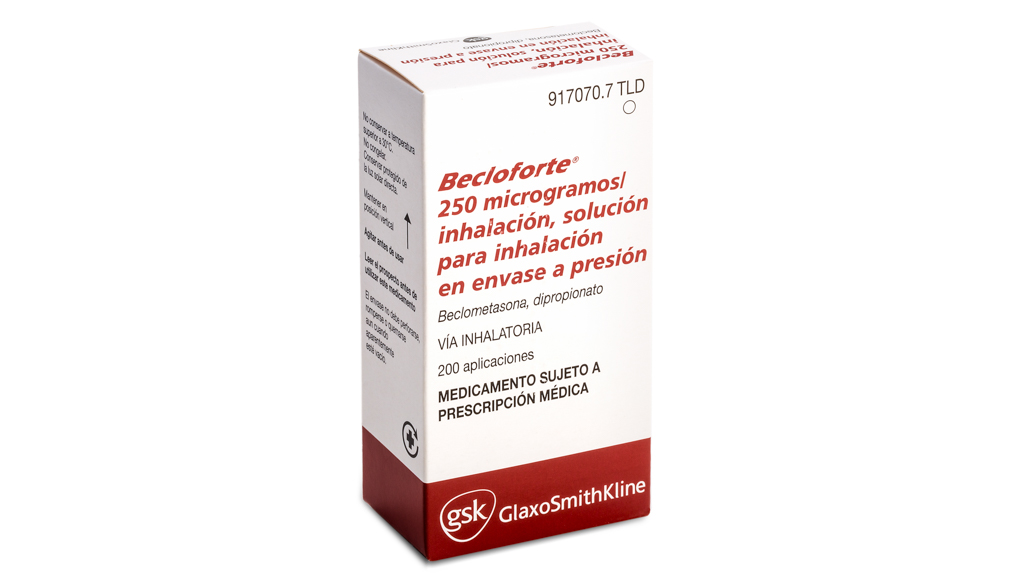
PHARMACEUTICAL DOSE FORMS
- INHALATION SOLUTION IN THE CONTAINER TO PRESSURE
ROUTES OF ADMINISTRATION
- INHALATION
STRENGTH
- 250 ΜG BECAOMETASONA DIPROPIONATE
ACTIVE INGREDIENTS
- BECLOMETHASONE DIPROPIONATE
EXCIPIENTS
- GLYCEROL
- ANHYDROUS ETHANOL
CHARACTERISTICS
- MEDICINAL PRODUCT SUBJECT TO MEDICAL PRESCRIPTION. LONG-TERM TREATMENT
- MEDICAL PRESCRIPTION
- FOR THE RESPIRATORY SYSTEM
ATCCODES
- R03B – OTHER AGENTS AGAINST DISEASES, OBSTRUCTIVE AIRWAY, INHALATIONAL
- R03BA – GLUCOCORTOCOIDES
- R03BA01 – BECLOMETASONE